Image copyright
Nicolas Rivron
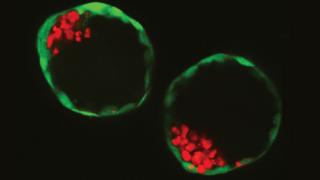
Two of the embryo-like structures grown by the scientists in the lab
Dutch scientists have built “synthetic” embryos in their laboratory using mouse cells other than sperm and eggs.
The stem cell breakthrough, described in Nature journal, is not for cloning people or animals, but about understanding why many pregnancies fail at an early stage – implantation.
The embryos, made in a dish, attached to the womb lining of live female mice and grew for a few days.
Studying the process could help human fertility, experts say.
Early miscarriage
Many miscarriages occur before a woman is even aware she is pregnant, when the fertilised egg does not manage to implant in the womb.
Experts do not fully understand why this occurs, although it is likely to be linked to abnormalities in the growing embryo.
Studying how early embryos develop is tricky both ethically and technically.
Using stem cells, rather than sperm and eggs, to make model embryos could offer a plentiful supply for scientific research.
Image copyright
Nicolas Rivron
Representation of the synthetic embryo
‘Large numbers’
Stem cells are immature cells that can turn into many different types in the body during early life and growth.
Dr Nicolas Rivron, from the Merlin Institute of Maastricht University, and his team created the embryo-like structures by mixing two types of stem cells from mice.
Under the microscope, they looked identical to real early embryos or blastocysts, with the same spherical ball of cells that would usually go on to make the placenta and baby.
The researchers were then able to observe it implant in mice – something that hasn’t been done before, although other scientists have created stem cell embryos for research.
Dr Rivron told the BBC: “We can now generate extremely large numbers of these embryos and study them in detail. It could help us understand why some embryos fail to implant and let us screen for drugs that might help with fertility.”
He said there were no plans yet to repeat the results using human stem cells. That would require approval.
Prof Robin Lovell-Badge, an expert at the UK’s Francis Crick Institute, said the prospects for obtaining human embryo-like structures in this way was currently “very remote”.
“This is a pity for basic research because it would be very useful to have a limitless supply of human blastocyst-like stage embryos to understand the relevant cell-cell interactions required to make normal embryos and to study mechanisms of implantation.
“However, it may come as a relief to others that such a method of producing many genetically identical human embryo-like structures that might be capable of implantation is not feasible – even if it would be illegal to implant them into women, as is clearly the situation in the UK.”
Dr Harry Leitch, Group Head at the MRC London Institute of Medical Sciences and Honorary Clinical Lecturer at Imperial College London, said it was the most successful attempt so far at building an early embryo from stem cell lines.
Dr Dusko Ilic, a stem cell expert at King’s College London, said: “This is the first time that scientists have been able to shed a light on the molecular mechanisms of implantation and these findings may help us to understand more about some aspects of infertility and improve outcomes of assisted reproduction.”



